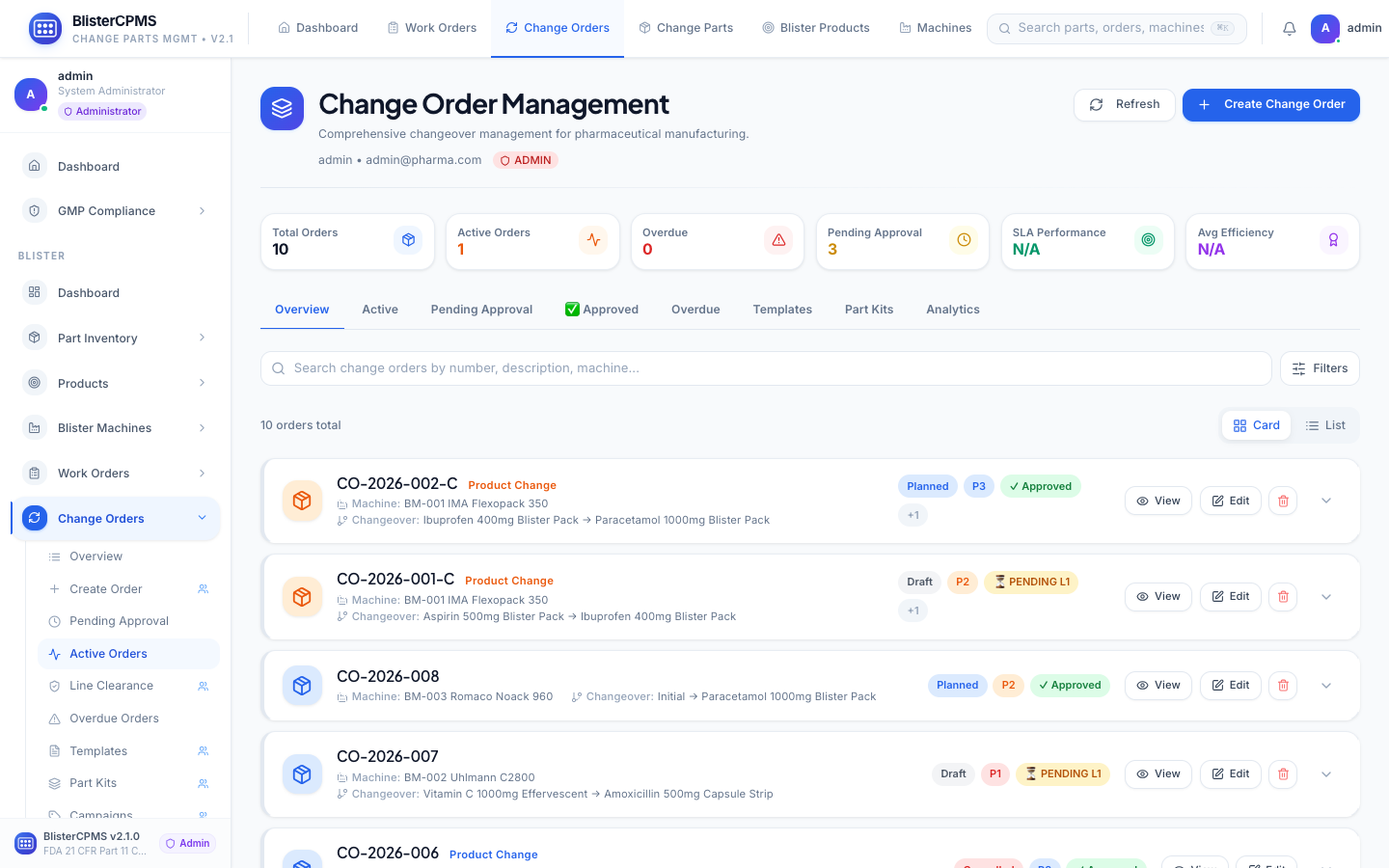
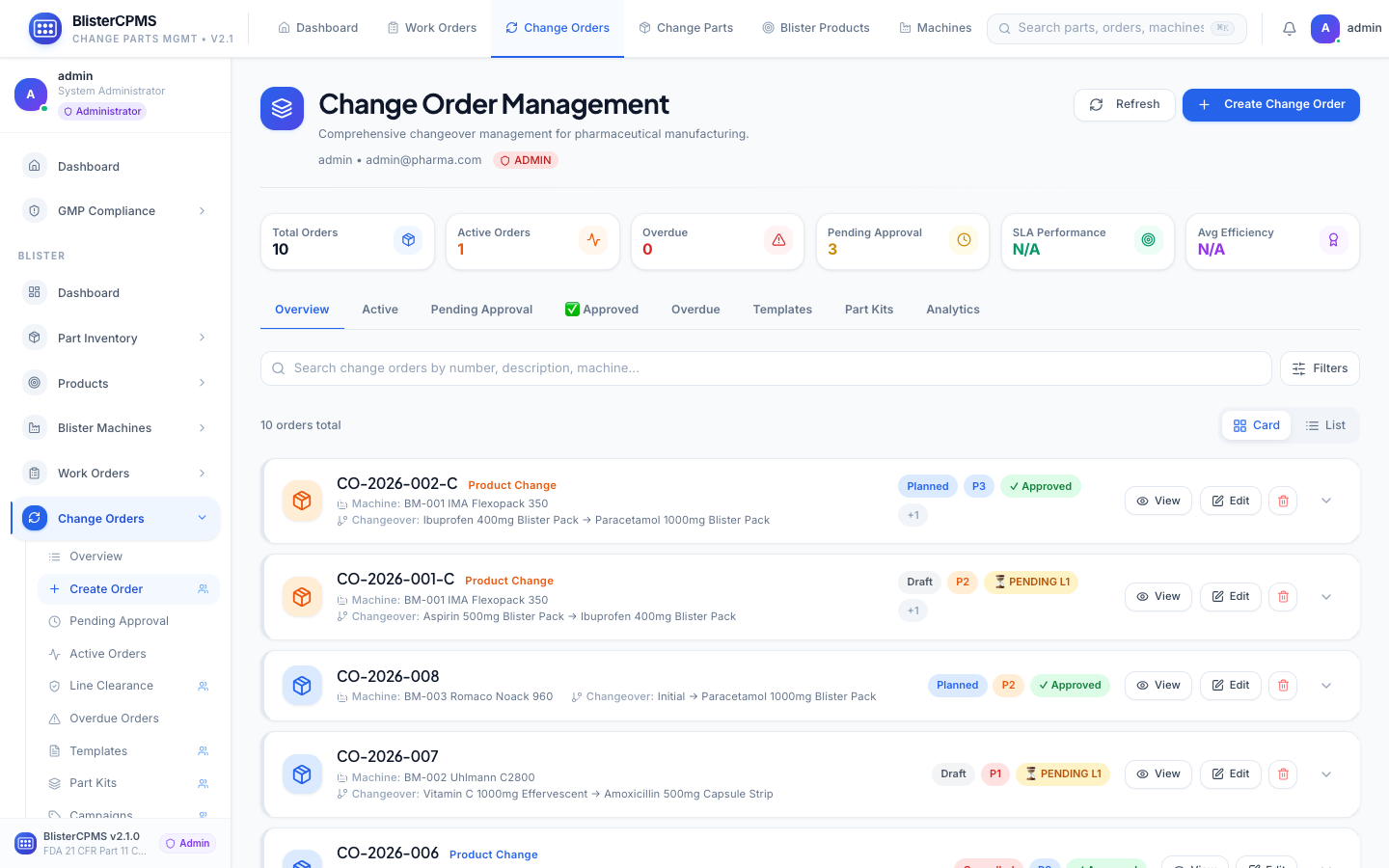
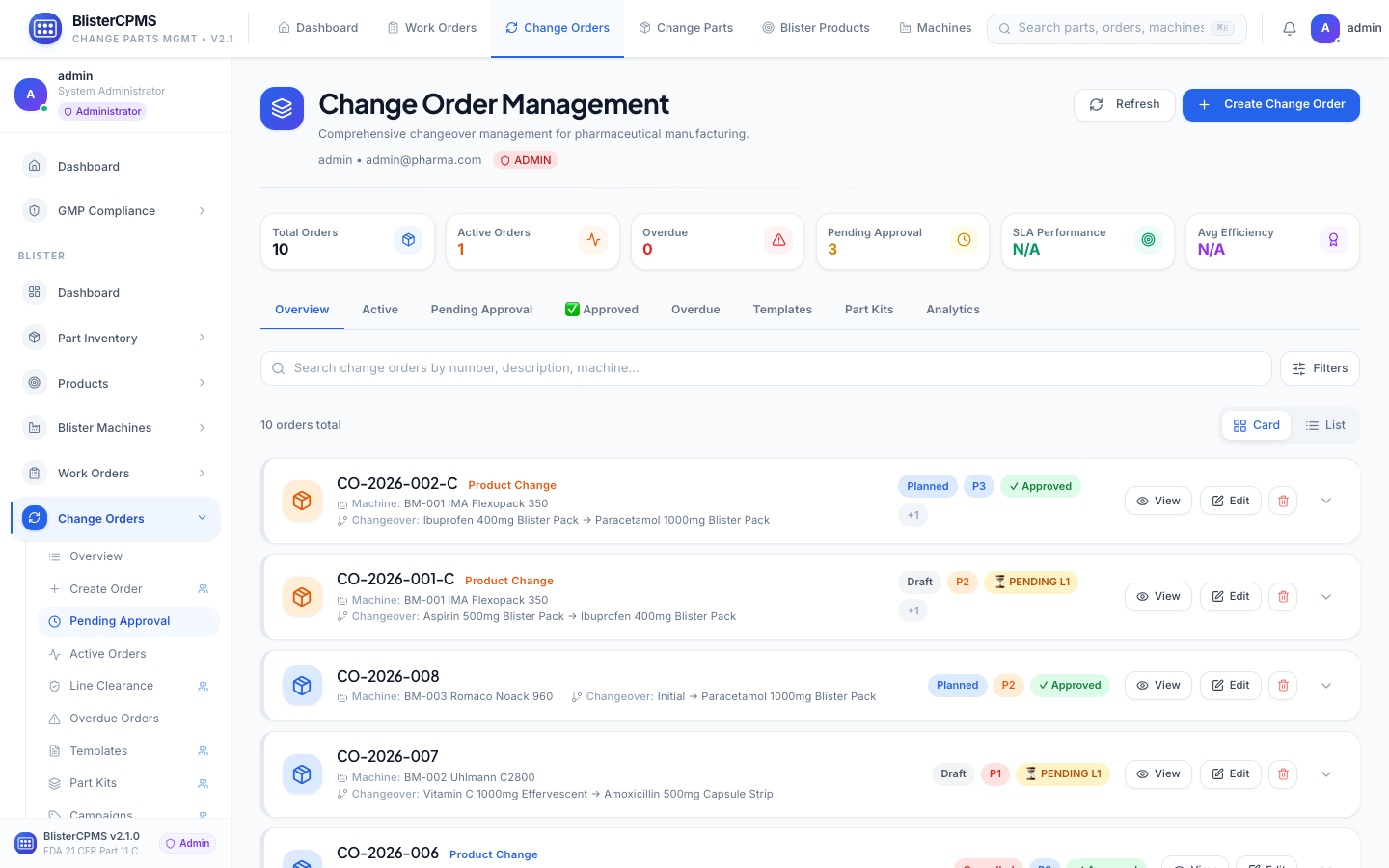
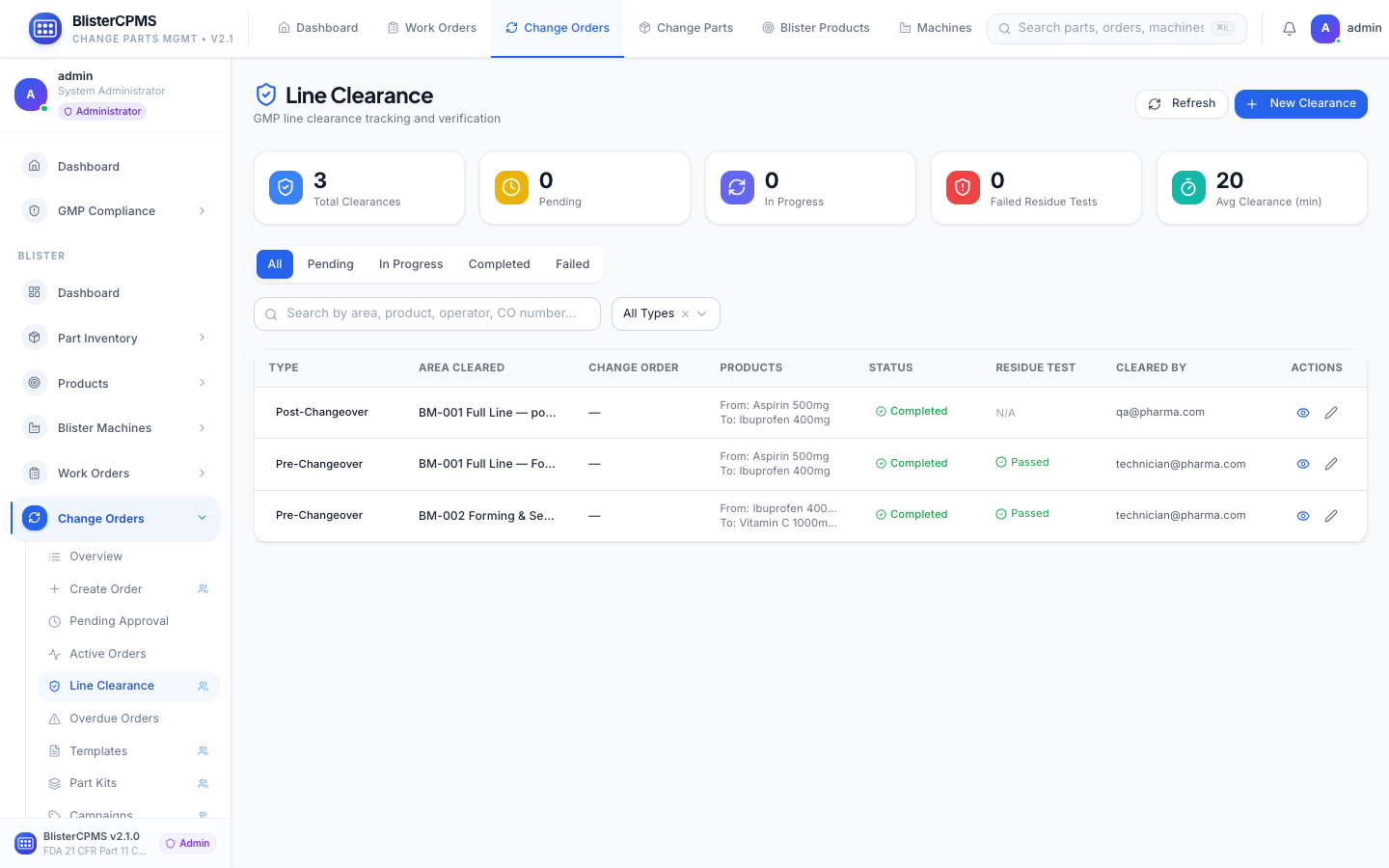
Change Orders
8-tab lifecycle: Active, Pending, Approved, Overdue, Templates, Part Kits, Analytics. Multi-stage approval with electronic signatures.
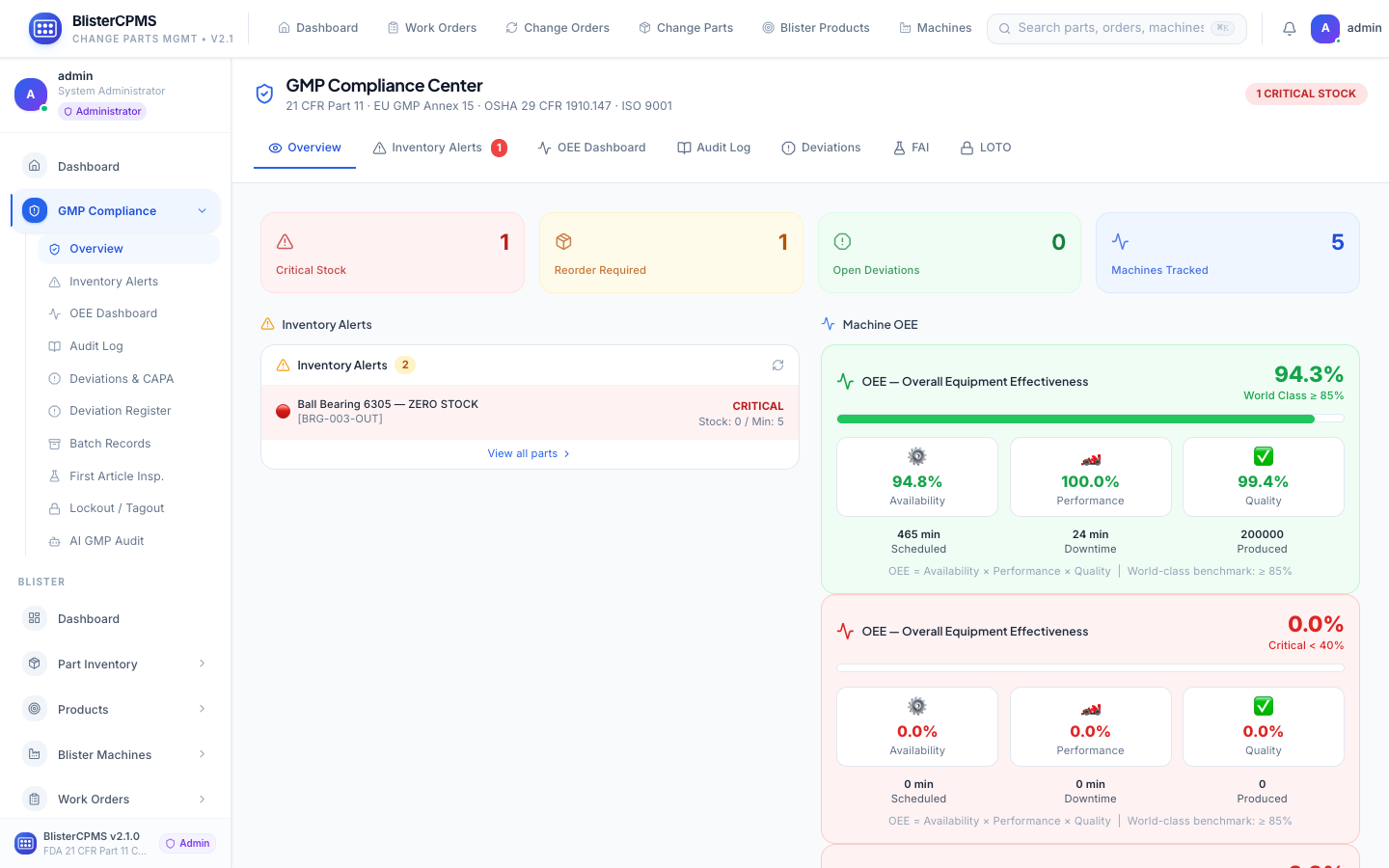
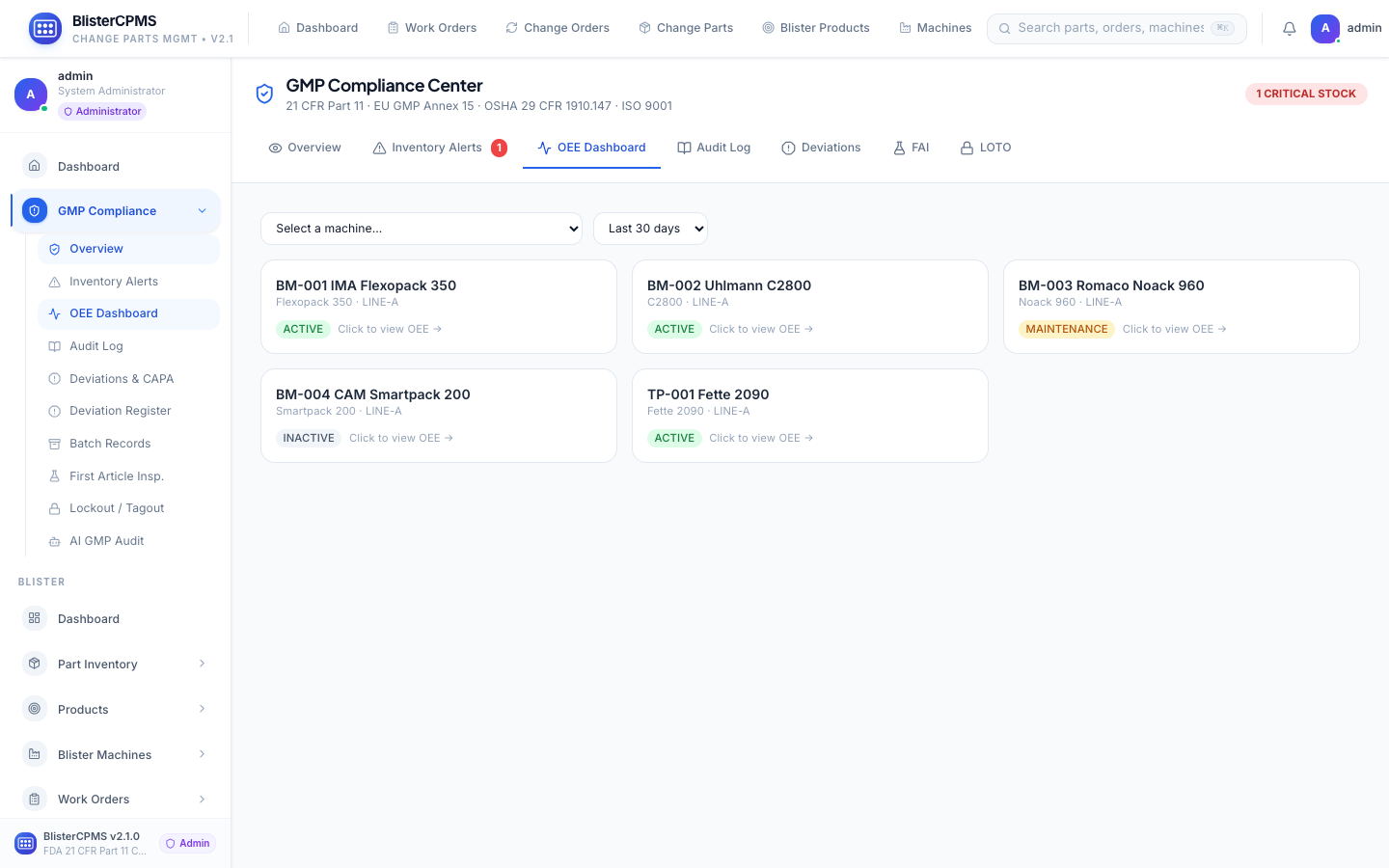
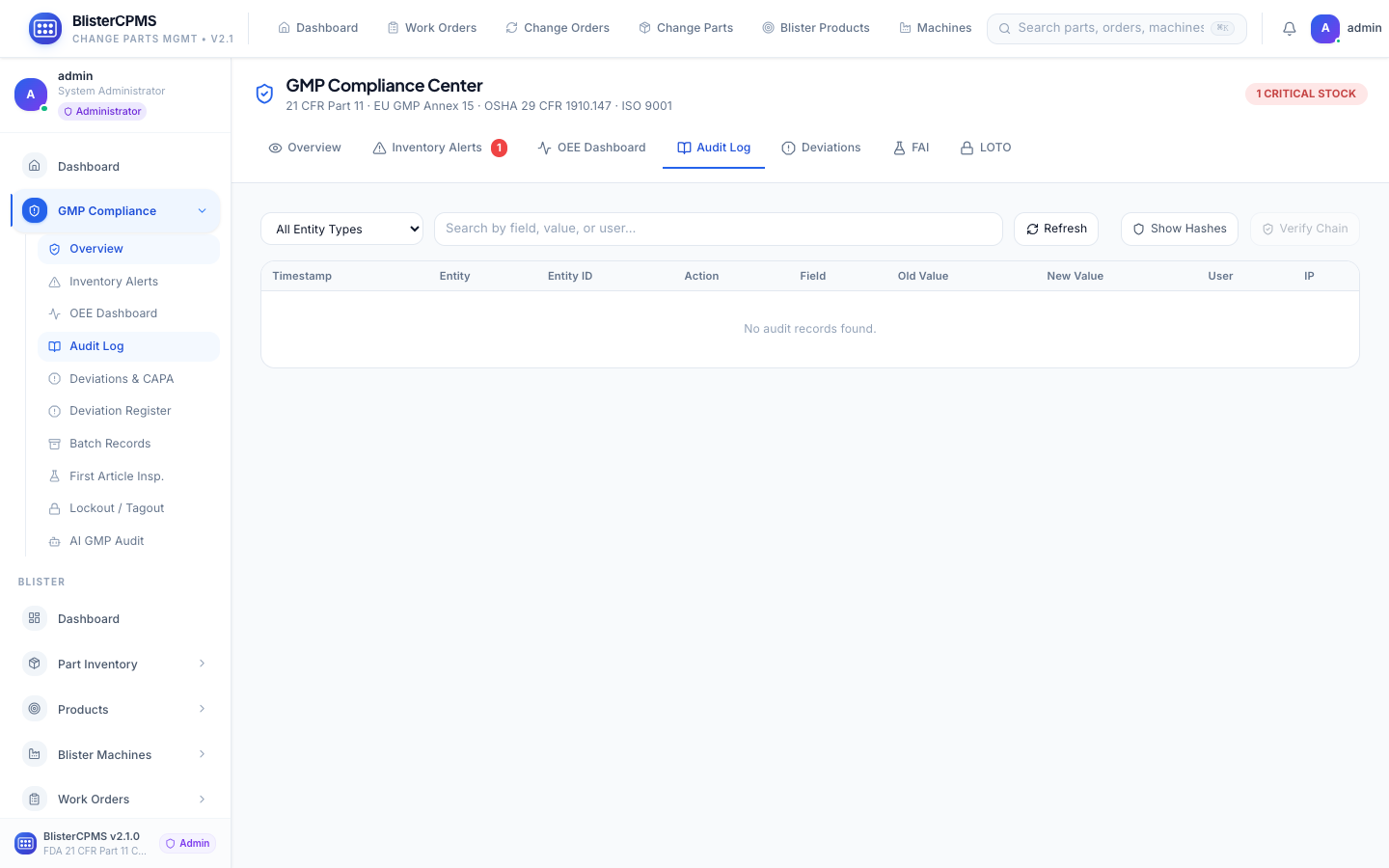
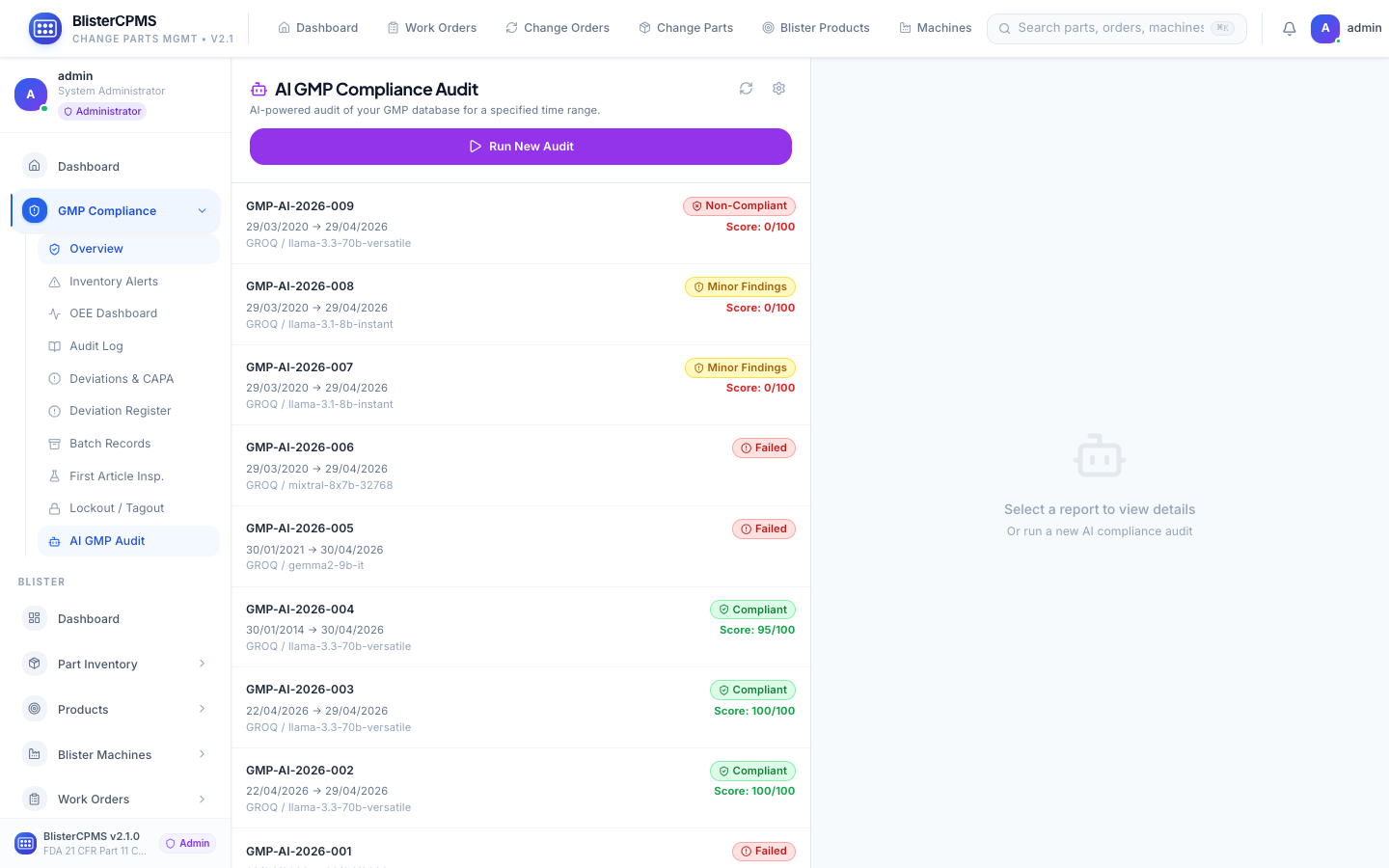
GMP Compliance
7 tabs: OEE, Audit Log, Deviations/CAPA, FAI, LOTO, Batch Records, AI Audit Scoring. Immutable 21 CFR Part 11 audit trail.
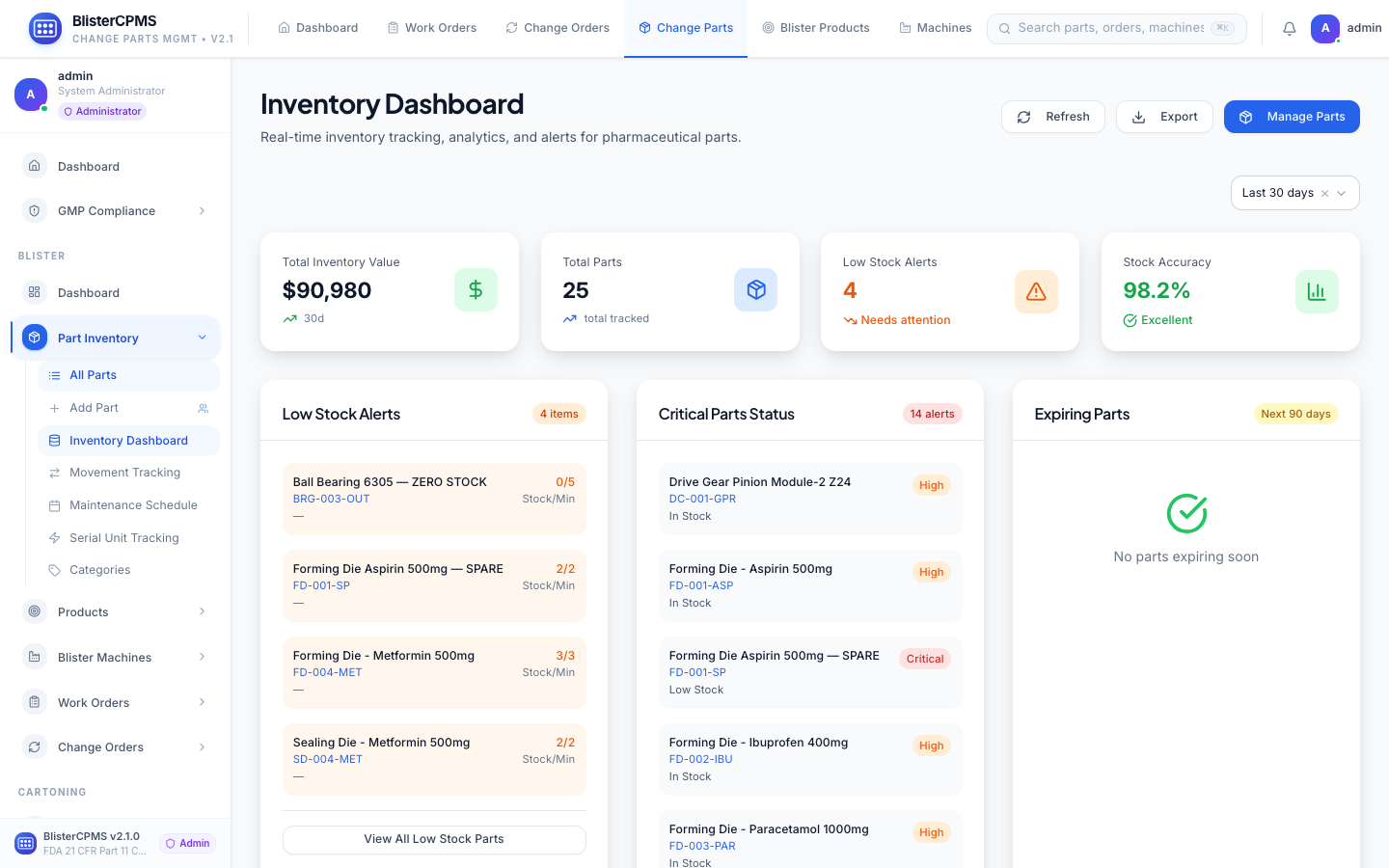
Parts Inventory
7-tab inventory with lot & serial tracking, expiry alerts, reorder automation, maintenance scheduling, and analytics.
Work Orders
Full work order lifecycle: create, assign, schedule, execute, close. Batch creation, template-driven flows, SLA tracking.
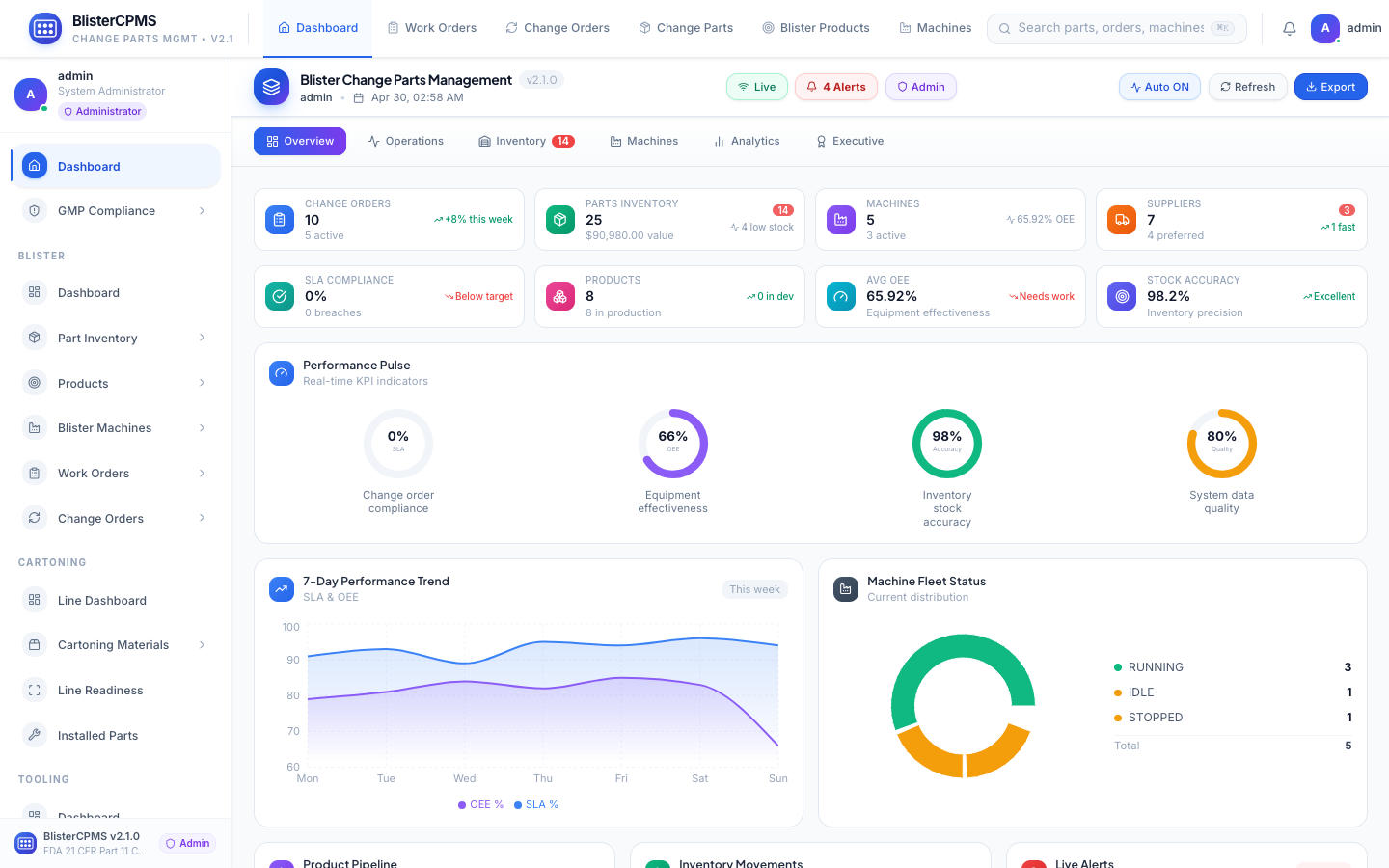
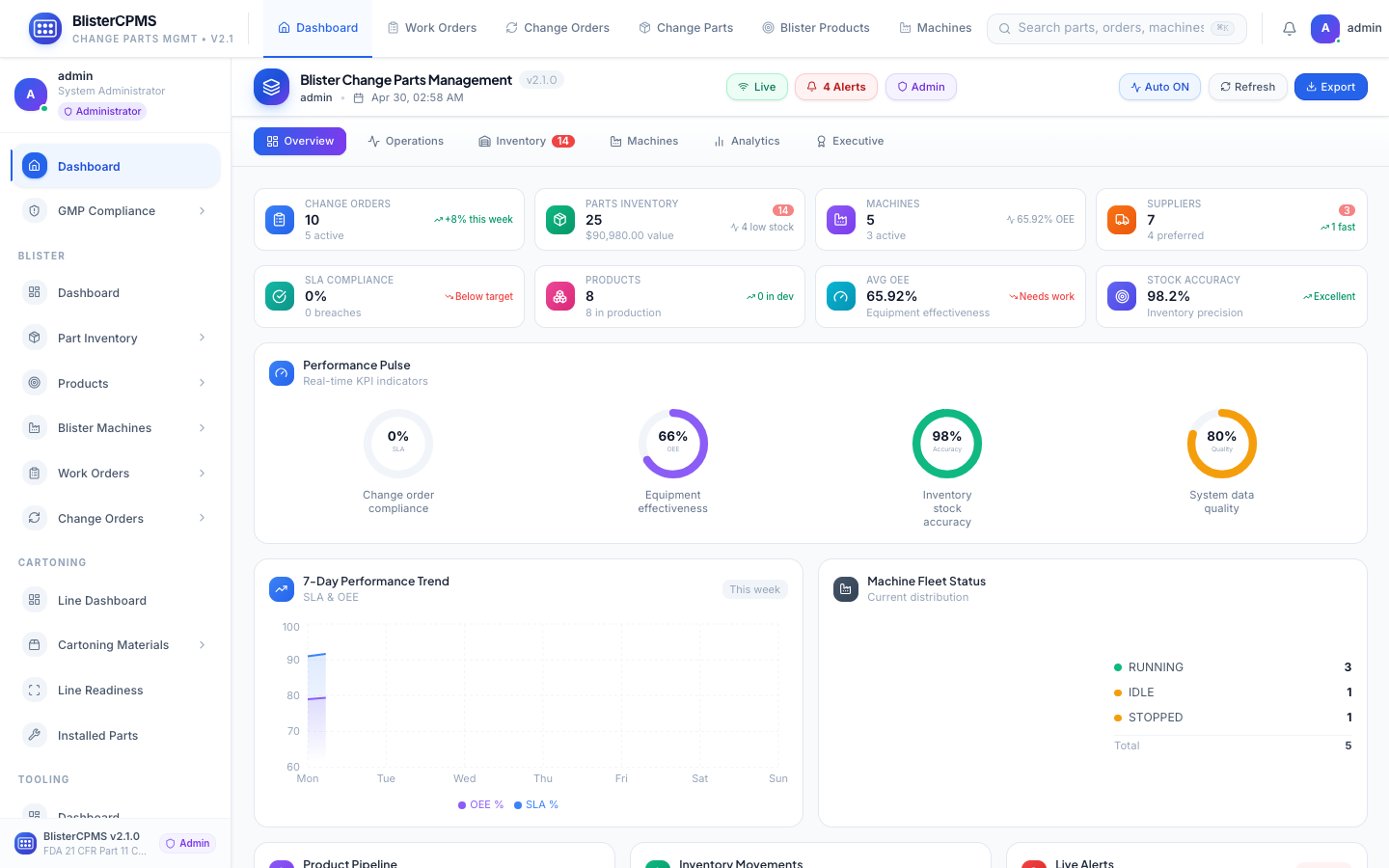
Global Dashboard
6 real-time views: Overview, Operations, Inventory, Machines, Analytics, Executive. One command centre.
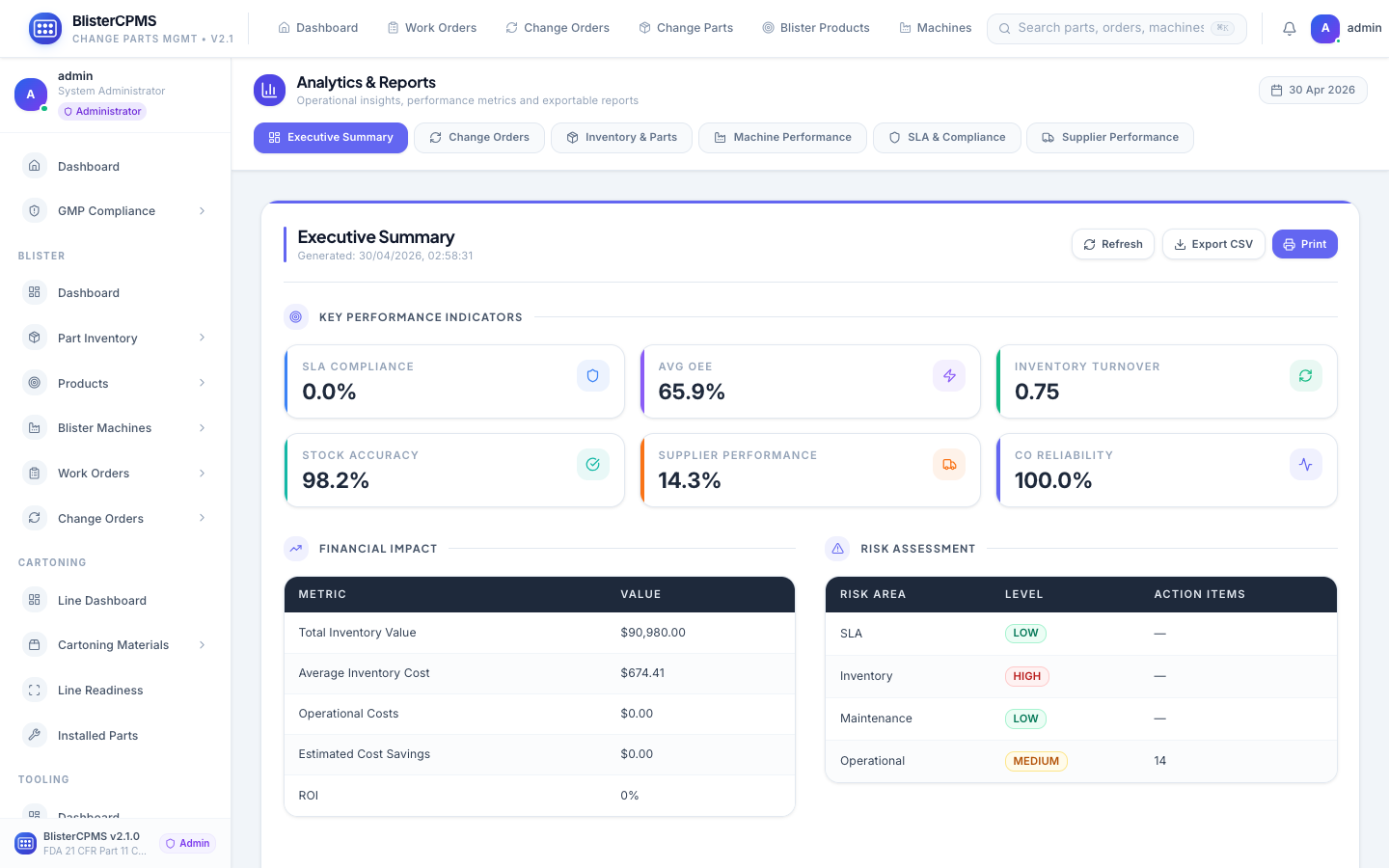
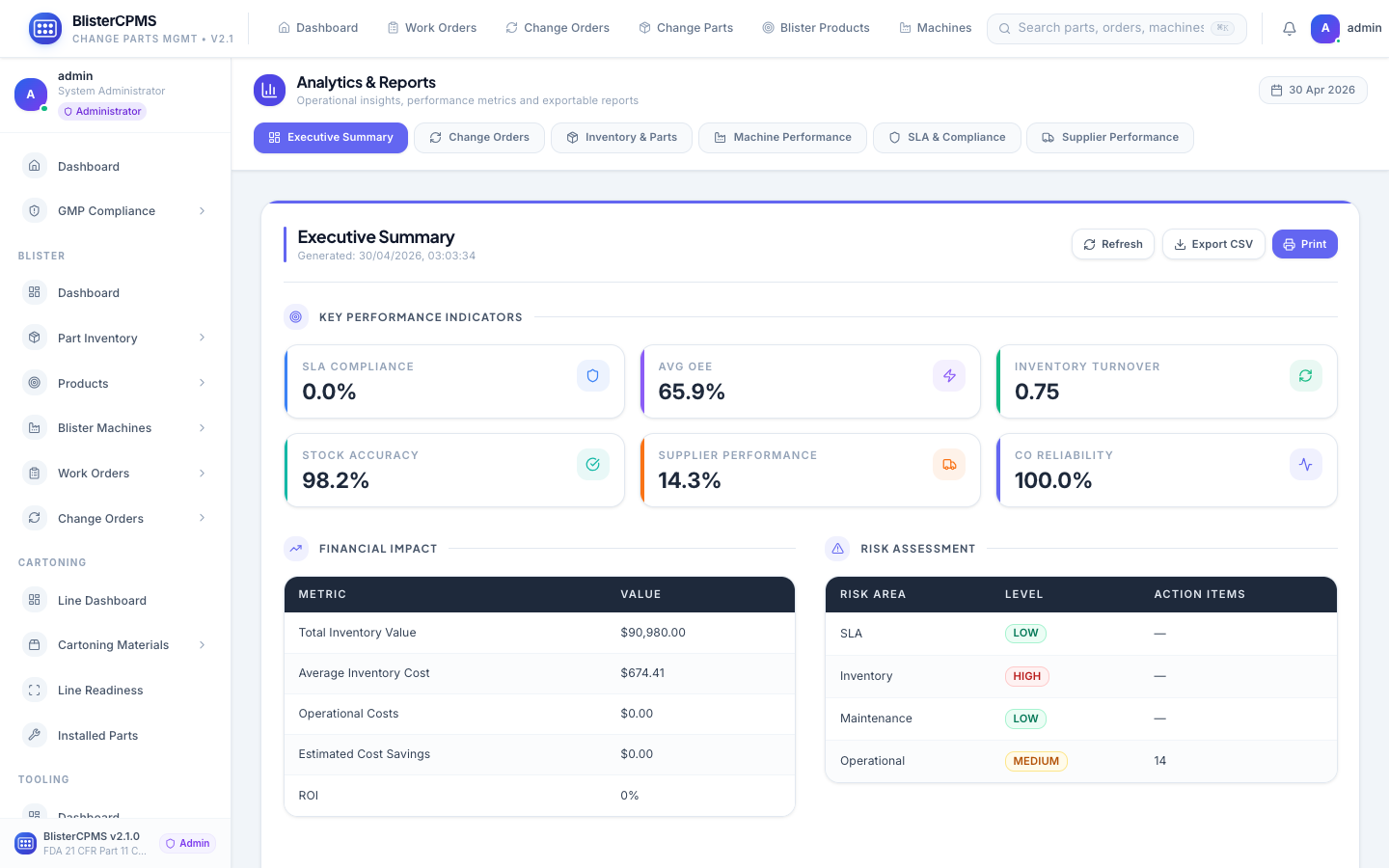
Analytics & Reports
6 report types: Executive, Change Orders, Inventory, Machine Performance, SLA & Compliance, Supplier Performance.
Warehouse & Locations
6-tab location management: Utilization, Hierarchy, Temperature zones, Capacity, Analytics. Multi-location tracking.
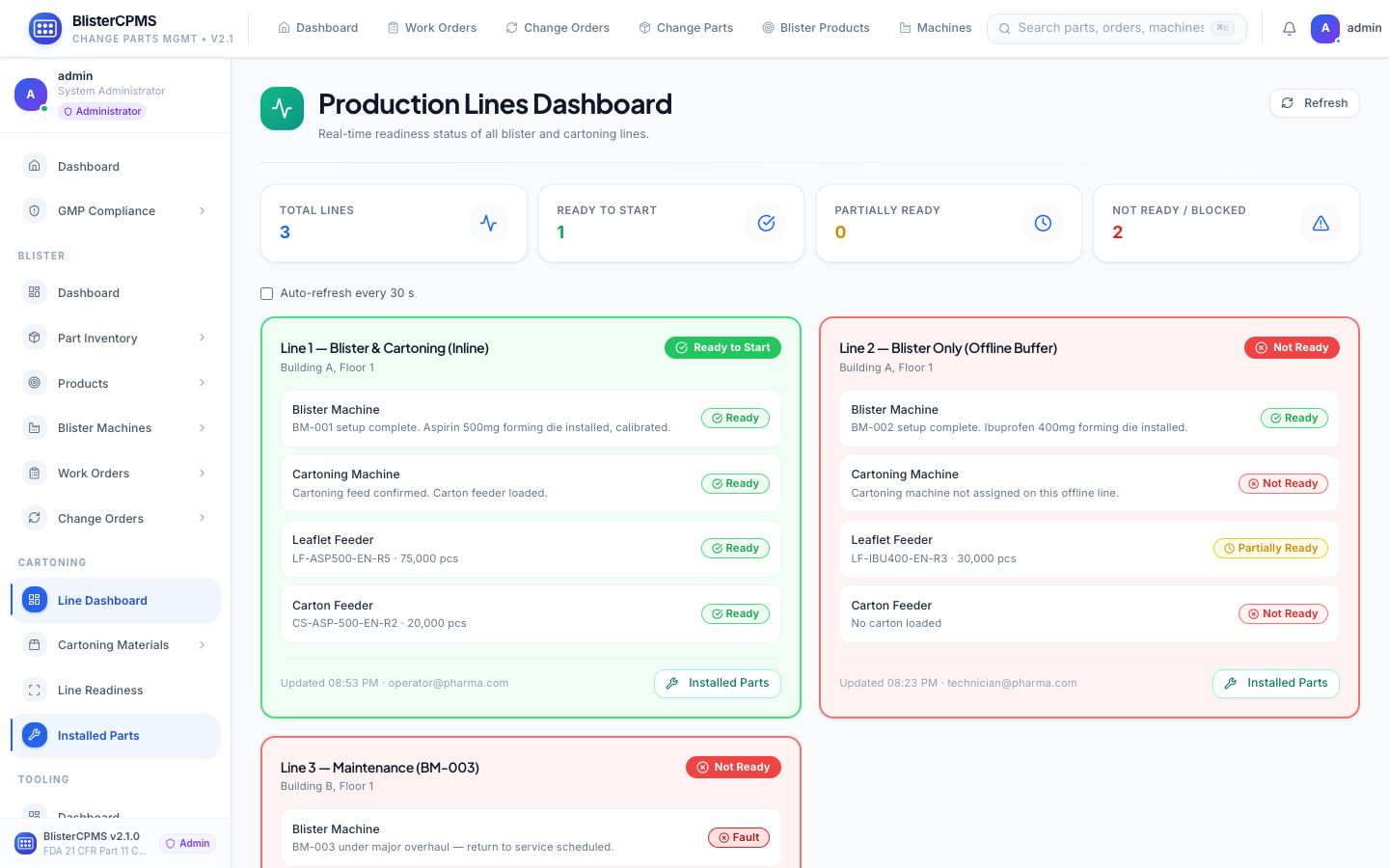
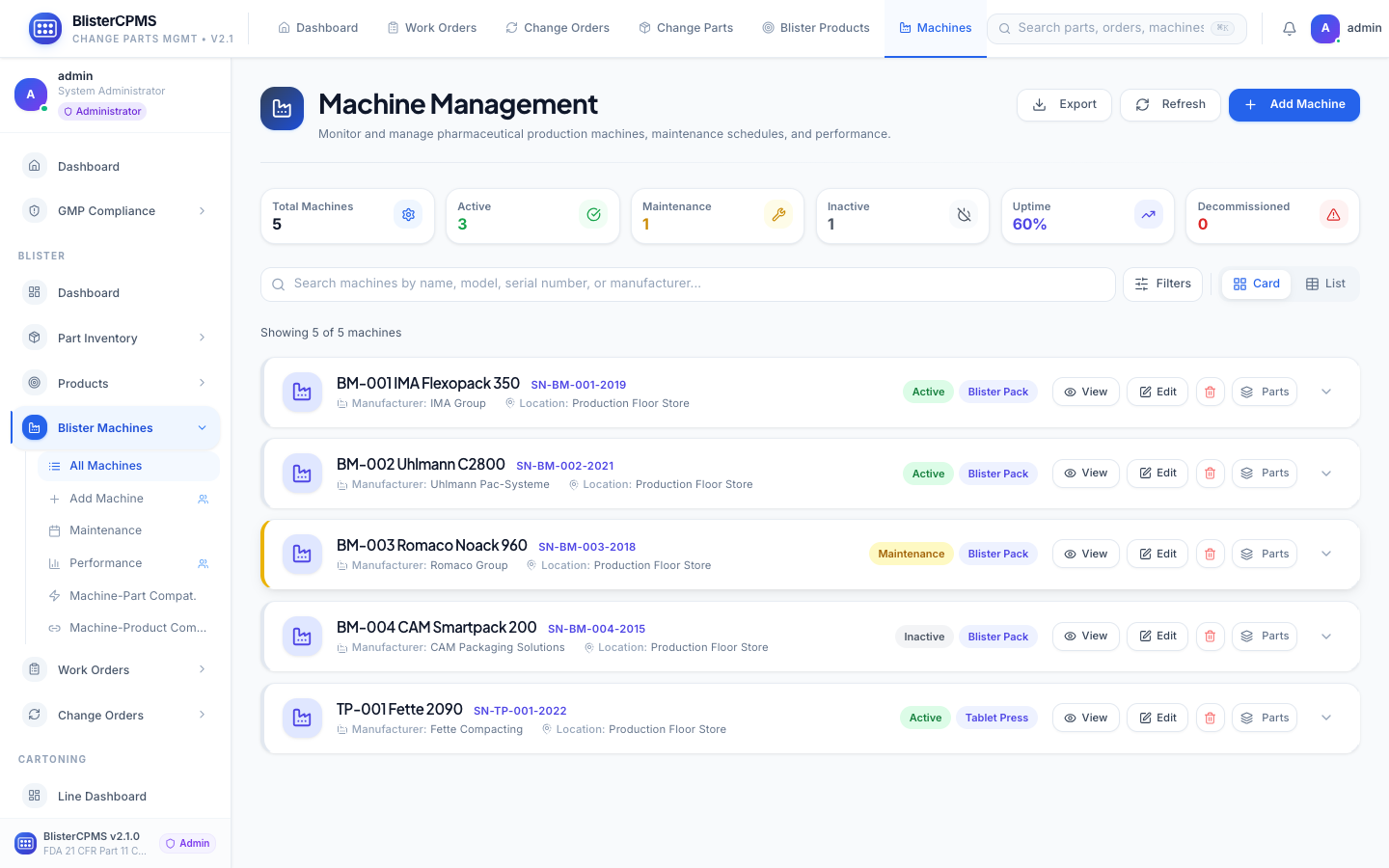
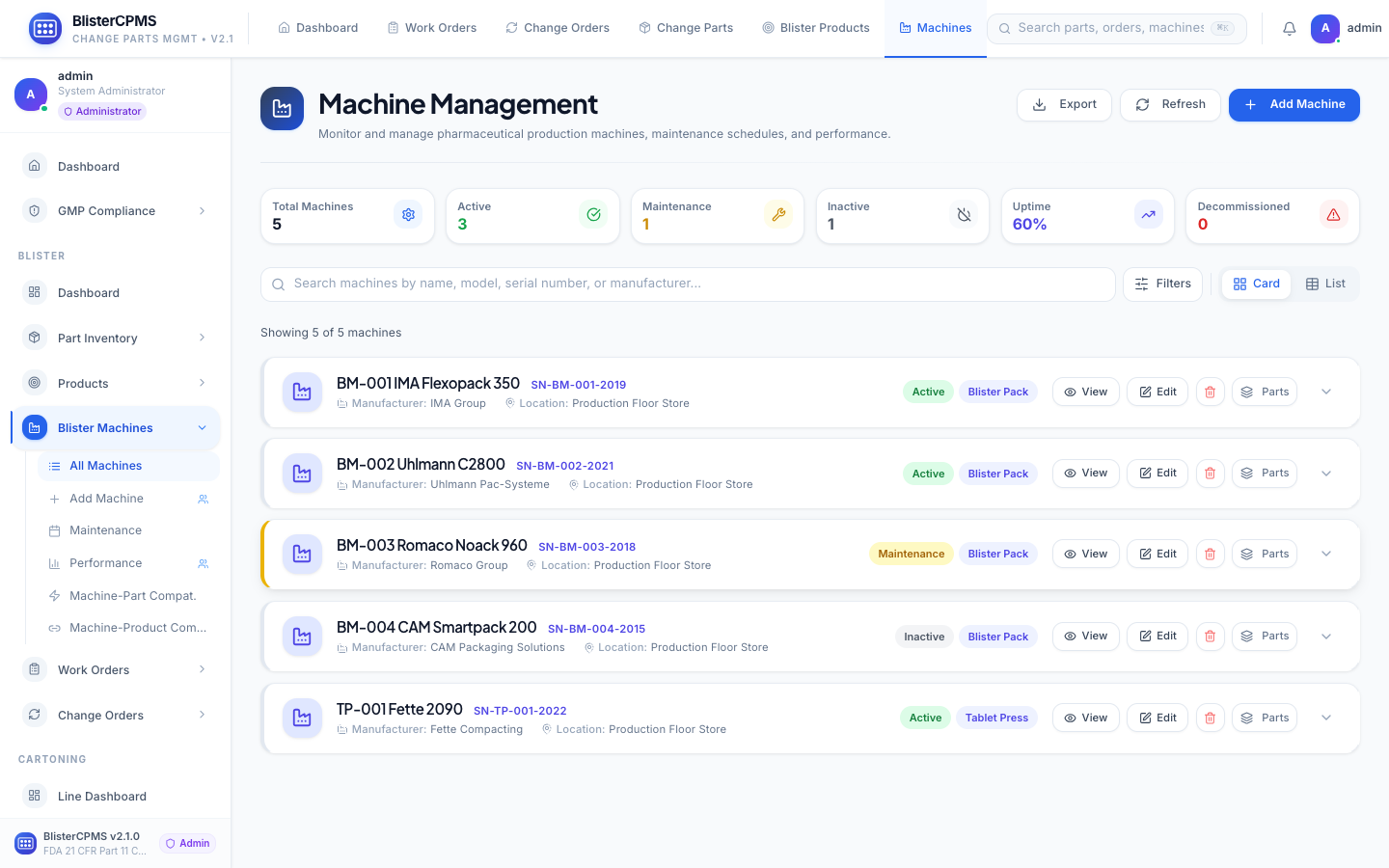
Blister Machines
Complete machine lifecycle: status monitoring, OEE trending, maintenance history, installed parts, benchmarking.
System Settings
19 configuration sections: users, roles, master data, AI scoring config, workflow rules, email templates, backup & recovery.